
Gretchen Hamn (L) and Margie Young screen a premature infant for retinopathy of prematurity. (Photos: Katherine C. Cohen)
We’re in the Neonatal Intensive Care Unit at South Shore Hospital. Six tiny, swaddled preemies are ready to be examined, their eyes numbed and their pupils dilated with special drops.
Gretchen Hamn, NNP, and medical assistant Margie Young go from isolette to isolette. Young tends to the first baby and gently positions him for his exam. Hamn pulls over a cart and extends a kind of hose with a camera at the tip. This she places directly on each of the baby’s eyes, taking a digital video of his retinas. Full story »
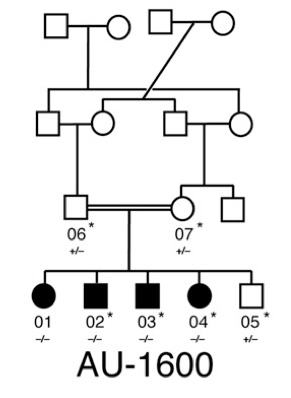
In this family with 4 children with autism, genetic mapping and whole-exome sequencing identified a mutation in SYNE1, a gene that's known but never before associated with autism.
Autism clearly runs in some families, yet few inherited genetic causes have been found. A major reason is that these causes are so varied that it’s hard to find enough people with a given mutation to establish a clear pattern. Now, three large Middle Eastern families with autism spectrum disorders (ASDs) have led the way to a few more mutations, potentially broadening the number of genetic tests available to families.
What’s fascinating is that the mutations, described earlier this week in Neuron, affect genes known to cause severe, often lethal genetic syndromes. Milder mutations in the same genes, found through genomic sequencing, primarily cause autism.
Researchers Tim Yu, MD, PhD, Maria Chahrour, PhD, and senior investigator Christopher Walsh, MD, PhD, of Boston Children’s Hospital, started with three large families that had two or more children with an ASD, in which the parents were first cousins. Cousin marriages are a common tradition in the Middle East that greatly facilitates the identification of inherited mutations—as does large family size. Full story »
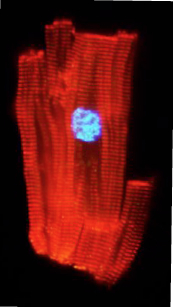
A heart muscle cell from an 8-year-old beginning the process of mitosis: The cell nucleus is preparing to divide. (Courtesy Bernhard Kühn)
For more than 100 years, people have been debating whether human hearts can grow after birth by generating new contractile muscle cells, known as cardiomyocytes. Recently, Bernhard Kühn, MD, at Boston Children’s Hospital and his colleagues added fuel to the debate—and hope for regenerative therapies for diseased hearts—with their findings that infants, children and adolescents are indeed capable of generating new cardiomyocytes.
Research in the 1930s and 1940s suggested that cardiomyocyte division may continue after birth, and recent investigations in zebrafish and newborn mice presented the possibility that some young animals can regenerate heart muscle through muscle cell division. Still, for many years, the accepted dogma among physicians and researchers was that human hearts grow after birth only through existing cells growing larger.
“This is a very sticky subject in cardiology,” says Kühn. Not only do long-held scientific beliefs die hard, but the ability to directly study heart cell growth in humans has been limited. “Healthy human hearts are hard to come by,” he says. Full story »
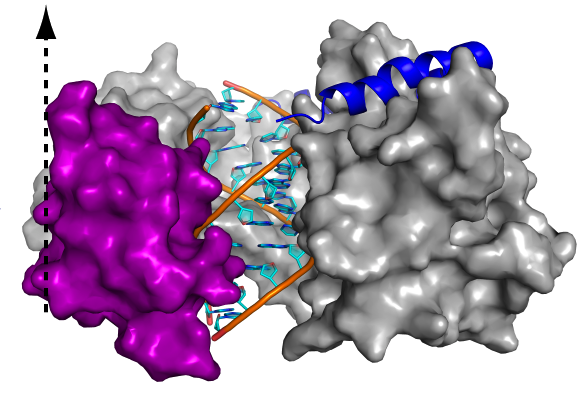
What you're looking at is one of the key ways in which our immune system recognizes viruses before they cause trouble: by sensing the physical presence of their genes. This image by Sun Hur, PhD, will help us better understand how.
Our immune system has immense powers of observation. It needs to in order to fend off the millions of bacteria, viruses, fungi, you name it, that we get exposed to every day.
I’m not talking about antibodies and T cells—parts of the immune system’s adaptive arm, which is fine-tuned to recognize a specific virus or bacterium. Rather, I’m talking about pattern recognition proteins—biological sensors capable of recognizing features and structures that only bacteria or viruses have. These make up the immune system’s innate arm, which essentially primes the body to attack anything that looks remotely like it doesn’t belong.
For instance, our cells carry sensors that can detect double-stranded RNA (dsRNA), which certain kinds of viruses use to encode their genome—like the rotavirus, which causes severe diarrhea in infants and small children. Our genome, by contrast, is encoded in DNA, and the RNA we make is single-stranded; if there’s dsRNA present, it means there’s a virus around.
In a recent paper in Cell, Sun Hur, PhD, of the Program in Cellular and Molecular Medicine at Boston Children’s Hospital, and one of her postdoctoral fellows, Bin Wu, PhD, spotlight one of our dsRNA pattern recognizers, a protein called MDA5. Full story »

(Garry Knight/Flickr)
Vector has been deliberating about its predictions for 2013, consulting its many informants. Here’s where we’re putting our money this year; if you have other ideas, scroll to the bottom and let us know.
Genome sequencing scaling up at health care institutions
Last year we predicted genome sequencing’s entry into the clinic; this could be the year it goes viral. Technology companies with ever-faster sequencers and academic medical centers are teaming up at a brisk pace to offer genomic tests to patients. Just in the past two weeks, a deal was announced between The Children’s Hospital of Philadelphia and BGI-Shenzhen to sequence pediatric brain tumors; Partners HealthCare and Illumina Inc. announced a network of genomic testing laboratories; Full story »
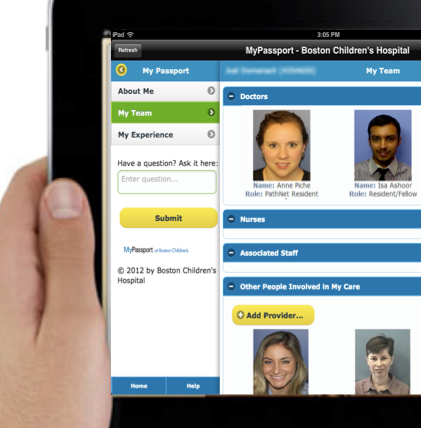 In just a 24-hour period, patients in the hospital typically see a variety of doctors, nurses, x-ray technicians and other medical professionals, and undergo a plethora of diagnostic tests—without an understanding of how all of it comes together to make them well.
In just a 24-hour period, patients in the hospital typically see a variety of doctors, nurses, x-ray technicians and other medical professionals, and undergo a plethora of diagnostic tests—without an understanding of how all of it comes together to make them well.
The Diversity and Cultural Competency Council (DCCC) at Boston Children’s Hospital recently conducted a three-year study on patient satisfaction. It found that the main reason patients were sometimes dissatisfied was because they felt unfamiliar with the medical information they were receiving, and had difficulty understanding who was part of their care team and how best to communicate with them. And so the idea of MyPassport was born. Full story »

A new spinoff business will make large-scale genomic diagnostics a reality in medical practice (Image: Rosendahl)
Genomic sequencing and molecular diagnostics are becoming a global business. At the recent American Society of Human Genetics meeting, dazzling technologies for reading genetic code were on display—promising faster, cheaper, sleeker.
Nevertheless, it’s become clear that the ability to determine someone’s DNA or RNA sequence doesn’t automatically translate into useful diagnostics or even actionable information. In fact, the findings are often confusing and hard to interpret, even by physicians.
That’s where academic-industry partnerships can flourish—tapping the deep expertise of medical research centers to bring clinical meaning to sequencing findings. Yesterday, Boston Children’s Hospital and Life Technologies Corp. announced a new venture with a great list of ingredients: fast, accurate, scalable sequencing technology—Life’s Ion Proton® Sequencer—but also research and clinical experience in rare and genetic diseases, bioinformatics expertise to handle the big data, and the medical and counseling expertise to create meaning from the results. Full story »

Inventions need a little extra incubating to make them attractive to health care.
Jenna Rose is director of Healthbox, a platform that brings together entrepreneurs, strategic partners, industry experts and investors to accelerate innovative healthcare solutions. She spoke recently at Boston Children’s Hospital at a forum sponsored by the Innovation Acceleration Program. She welcomes inquiries from entrepreneurs and others at [email protected].
When we think about the future of health, it’s generally medical science that captures our imagination—the source of groundbreaking pharmaceuticals, medical devices and diagnostics. But what about the business of health care? With the passage of the Affordable Care Act and the widespread adoption of mobile technologies, there has never been a better time to be a health tech entrepreneur. One recent report suggests that the healthcare IT sector could receive more than $1B in venture capital in 2012.
But change won’t be easy. As they seek to disrupt this $2.7 trillion industry, health tech entrepreneurs face a unique set of challenges. Full story »
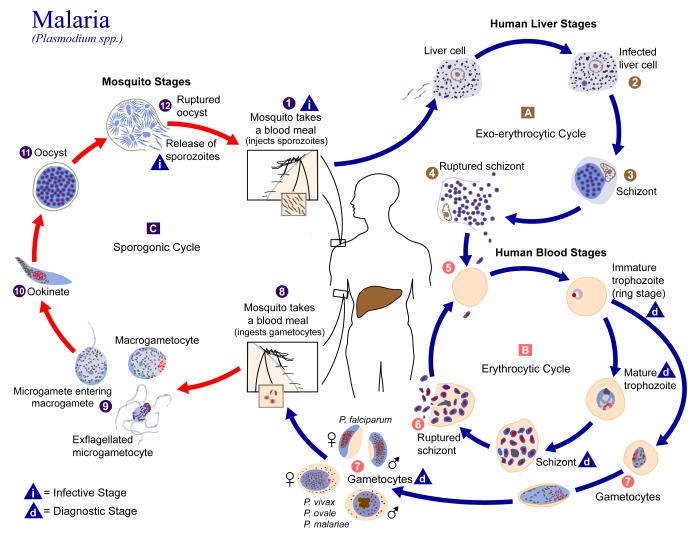
The exceedingly complex life cycle of malaria. Within it lies the key to developing a vaccine against the parasite. (CDC)
The malaria parasite (or parasites: four species of Plasmodium can cause malaria in people) has a really complex life cycle. That complexity has allowed this mosquito-borne parasite from bringing untold misery to the human race for millennia. The World Health Organization thinks it causes 216 million cases of disease every year, while the U.S. Centers for Disease Control and Prevention estimates that some 3.3 billion people live at risk of malaria infection around the globe. Even in the United States, where malaria was officially eradicated 60 years ago, there are still about 1,500 cases every year.
All these numbers add up to one fact: we need a vaccine, badly. This is where malaria’s complexity becomes a problem. Full story »

What will happen to medically complex children if insurance coverage is reduced and fewer pediatricians are trained to care for them? (Image: Wikimedia Commons)
Jay Berry, MD, MPH, is a pediatrician and hospitalist in the Complex Care Service at Boston Children’s Hospital. His most recent research appears in the Archives of Pediatrics & Adolescent Medicine, accompanied by editorials on the findings’ implications for health care and residency training. Berry further discusses its implications in this podcast.
My first encounter with a children’s hospital was as a first grader in 1980, when my 5-year-old cousin was diagnosed with cancer. Although her family was challenged to afford her cancer treatments, St. Jude Children’s Hospital in Memphis welcomed her and treated her cancer into remission. I remember my parents saying, “Everybody in that hospital loves children. No child is turned away.”
In 1997, walking into the Children’s Hospital of Alabama as a medical student, I felt the same sense of hope and courage. Everyone on the staff believed that they could make a difference in the lives of the children and families, despite the horrific illnesses that many of the children endured. I knew, immediately, that I wanted to become a pediatrician and to learn how to care for sick children. Full story »

















