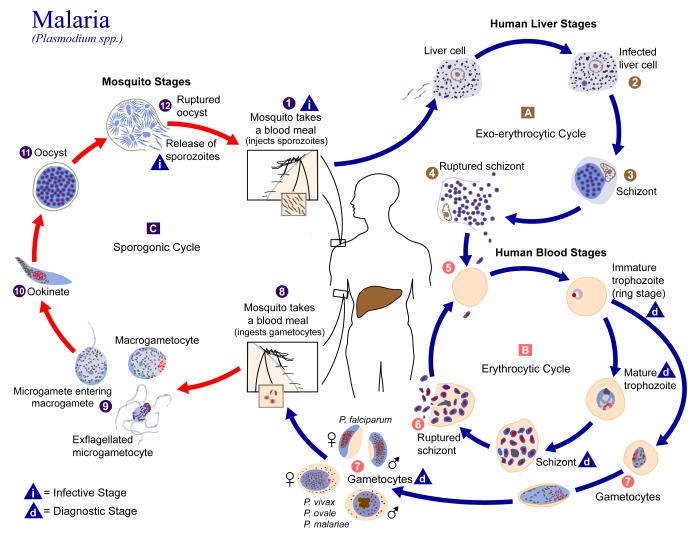
The exceedingly complex life cycle of malaria. Within it lies the key to developing a vaccine against the parasite. (CDC)
The malaria parasite (or parasites: four species of Plasmodium can cause malaria in people) has a really complex life cycle. That complexity has allowed this mosquito-borne parasite from bringing untold misery to the human race for millennia. The World Health Organization thinks it causes 216 million cases of disease every year, while the U.S. Centers for Disease Control and Prevention estimates that some 3.3 billion people live at risk of malaria infection around the globe. Even in the United States, where malaria was officially eradicated 60 years ago, there are still about 1,500 cases every year.
All these numbers add up to one fact: we need a vaccine, badly. This is where malaria’s complexity becomes a problem.
The parasite’s surface proteins—and available vaccine targets—change with each step in its life cycle, meaning that a vaccine against trophozoites won’t work against schizonts.
There is a promising pair of vaccine targets being investigated now. One of these is TRAP, a protein that helps the parasite move around in its sporozoite stage (the form passed from mosquitoes to people). But even there we have an obstacle. While we know the sequence of amino acids that makes up TRAP, we’ve never completely understood its shape, which can greatly influence how the body reacts upon seeing it both in a vaccine and on the real thing.
A team led by Timothy A. Springer, PhD, of Boston Children’s Program in Cellular and Molecular Medicine, used X-ray crystallography to work out both the three-dimensional structure of TRAP and how it changes shape as it helps the parasite move about. This information, reported in the Proceedings of the National Academy of Sciences, could help vaccine developers use TRAP to teach the immune system to attack malaria parasites when it sees them.
“There has been a misunderstanding in the field that TRAP is not immunogenic. However, we find that TRAP is highly immunogenic when it is correctly folded,” says Springer, who earlier this year analyzed the crystal structure of another major sporozoite protein, circumsporozoite, and found that a portion of it could be vulnerable to immune attack. “We will soon be testing TRAP vaccines in animals for complete protection against infection.
“Since TRAP is required for migration of sporozoites in the human body and for the first step in infection of liver cells in malaria,” he continues, “we are highly optimistic about its efficacy as a vaccine target, either alone or combined with circumsporozoite protein.”







