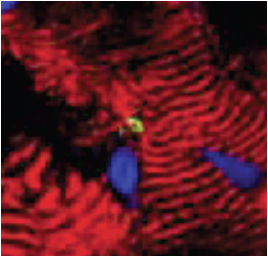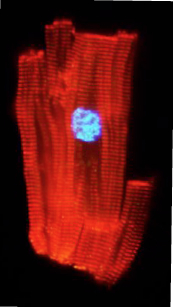
A heart muscle cell from an 8-year-old beginning the process of mitosis: The cell nucleus is preparing to divide. (Courtesy Bernhard Kühn)
For more than 100 years, people have been debating whether human hearts can grow after birth by generating new contractile muscle cells, known as cardiomyocytes. Recently, Bernhard Kühn, MD, at Boston Children’s Hospital and his colleagues added fuel to the debate—and hope for regenerative therapies for diseased hearts—with their findings that infants, children and adolescents are indeed capable of generating new cardiomyocytes.
Research in the 1930s and 1940s suggested that cardiomyocyte division may continue after birth, and recent investigations in zebrafish and newborn mice presented the possibility that some young animals can regenerate heart muscle through muscle cell division. Still, for many years, the accepted dogma among physicians and researchers was that human hearts grow after birth only through existing cells growing larger.
“This is a very sticky subject in cardiology,” says Kühn. Not only do long-held scientific beliefs die hard, but the ability to directly study heart cell growth in humans has been limited. “Healthy human hearts are hard to come by,” he says.
Kühn and his team, however, were able to study a large number of healthy hearts, securing samples from 28 unused donor hearts and 8 recently deceased persons’ hearts. The hearts ranged in age from 0 to 59 years. Using a new method for isolating individual cardiomyocytes, the team documented that these cells were still dividing after birth and significantly expanding their numbers. Rates of cell division were highest during infancy and then began a gradual decline, but cardiomyocytes kept dividing up until around age 20.
This research, recently published in the Proceedings of the National Academy of Sciences, offers the strongest evidence to date that proliferation of cardiomyocytes contributes to growth in healthy young human hearts—challenging the accepted wisdom and creating, as Kühn says, a “cellular blueprint for how the human heart grows after birth.”
What’s more, it raises the possibility of stimulating production of new cells to repair injured hearts, which could lead to new and better approaches for treating diseases of the heart muscle (cardiomyopathy) and heart failure in children. Using the same methods, researchers may continue to examine how heart muscle cells divide in people with heart disease and find ways to use this knowledge to create new treatments for children with heart defects.
“A lot more work needs to be done,” Kühn cautions, but he is excited to see where this work may lead.