“Sometimes it is the simplest things in medicine that stay the same over time,” says Liu, whose invention will be featured later this week at Boston Children’s Hospital’s Global Pediatric Innovation Summit + Awards 2014 (October 30-31). Full story »
From the category archives:
Devices
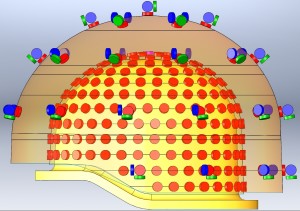
This array of sensors surrounding a baby's head will give researchers and eventually clinicians a high-resolution image of neural activity.
For years, that kind of thinking has been the domain of dreams. Little is known about infant brains, largely because sophisticated neuroimaging technology simply hasn’t been designed with infants in mind. Boston Children’s Hospital’s Ellen Grant, MD, and Yoshio Okada, PhD, are debuting a new magnetoencephalography (MEG) system designed to turn those dreams into reality. Full story »

With initial help from her mother, Kailee West, 6, quickly masters the basics of Puddingstone Place, an interactive virtual environment that helps children with autism develop language skills.
In the 1990s, Facilitated Communication (FC), in which assistants “facilitate” the typing of thoughts by minimally verbal children by supporting their hands, began raising hopes in the autism community. The unproven procedure caught fire, and Syracuse University established a nationally recognized Facilitated Communication Institute.
Upon closer examination, though, doubts emerged. The messages were surprisingly sophisticated and written by children who often were not even looking at the keyboard. Critics charged that the words were actually those of the facilitator rather than the patient. Studies and organizations began discrediting FC. Full story »
Fitbit, Jawbone, Nike, Withings…a lot of companies are already in the wearable/mobile health technology and data tracking game. But a couple of really big players are stepping on to the court.
At their most recent Worldwide Developers Conference, Apple announced both an app and a framework—Health and HealthKit—that will tie in with various wearable technologies and health apps. HealthKit will also feed data into electronic medical record (EMR) systems like Epic, which runs at some of the largest hospitals in the country. And rumors abound that an upcoming Apple smartwatch (iWatch? iTime? Only Tim Cook knows right now) will carry a host of sensors for tracking activity and health data.
Google also wants to get into the game with a health data framework called Fit that they announced at their I/O conference in June. Unlike Apple, its strategy seems more focused on providing a standard way for trackers, devices and apps from different manufacturers to talk to Android Wear devices.
What will entry of these big players mean? We asked Michael Docktor, MD, clinical director of Boston Children’s Hospital’s Innovation Acceleration Program. Full story »
Geoffrey Horwitz, PhD, is a business development associate in the Technology and Innovation Development Office (TIDO) at Boston Children’s Hospital. Follow him on Twitter @GeoffHorwitz
At the recent 2014 Biotechnology Industry Organization (BIO) International Convention, the message was clear: Digital health is the new blockbuster. For the first time ever, BIO spotlighted digital health, with a specific focus on how digital health is influencing the pharmaceutical and biotech industries. Also featured was a digital health zone where companies and other exhibitors from all over the world could demo their products and services to thousands of attendees.
In pharmaceutical lingo, a blockbuster is a drug that generates revenues of at least $1 billion. Digital health certainly fits this definition. By 2018, reports suggest that revenues will exceed $6 billion for wearable wireless devices alone. A recent McKinsey study found that 75 percent of consumers surveyed, of various ages and located throughout the world, would like to use digital health devices. Full story »
 Projections that the global mobile health market will boom to nearly $50 billion have ignited interest among innovators. A pair of physician innovators from Boston Children’s Hospital peg wearables as the technology to watch and offer a sneak peek at what adoption might mean, while others ask about the pediatric market for wearables and point to a few potential stumbling blocks. Read on for their views. Full story »
Projections that the global mobile health market will boom to nearly $50 billion have ignited interest among innovators. A pair of physician innovators from Boston Children’s Hospital peg wearables as the technology to watch and offer a sneak peek at what adoption might mean, while others ask about the pediatric market for wearables and point to a few potential stumbling blocks. Read on for their views. Full story »