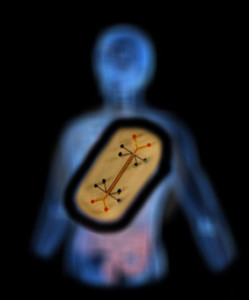
“Sometimes it is the simplest things in medicine that stay the same over time,” says Liu, whose invention will be featured later this week at Boston Children’s Hospital’s Global Pediatric Innovation Summit + Awards 2014 (October 30-31). Full story »
From the category archives:
Innovators’ stories
Magnetic needle tackles suturing in challenging surgical fields
by Joshua Feblowitz on October 27, 2014
Related posts:
Hacking Pediatrics 2.0 in tweets
by Nancy Fliesler on October 21, 2014
Parents, clinicians, app developers, designers and more had 18 hours to prototype digital healthcare solutions at Hacking Pediatrics, produced by Boston Children’s Hospital and MIT Hacking Medicine. To accompany our earlier post, we created this Storify. Full story »
Related posts:
- Hacking pediatrics: Improving the patient experience
- Hacking Pediatrics 2.0 draws 22 digital health pitches
- Creating a culture of crowd-sourced innovation: A look back at Hacking Pediatrics
- Hacking a solution for hydrocephalus… just not the one expected
- Bringing everyone to the table: Hackathon brings a clinician’s dietary innovation to life
Hacking Pediatrics 2.0 draws 22 digital health pitches
by Nancy Fliesler on October 21, 2014
The hackathon, produced by Boston Children’s Hospital in collaboration with MIT Hacking Medicine, brought out many common themes: Helping kids with chronic illnesses track their symptoms, take their meds and avoid lots of clinic visits. Helping parents coordinate their children’s care and locate resources. Helping pediatric clinicians make better decisions with the right information at the right time.
Hackathons have a simple formula: Pitch. Mix. Hack. Get Feedback. Iterate. Repeat—as many times as possible. Full story »
Related posts:
- Hacking pediatrics: Improving the patient experience
- Creating a culture of crowd-sourced innovation: A look back at Hacking Pediatrics
- Hacking a solution for hydrocephalus… just not the one expected
- Google and Apple enter digital health. Will they rocket-launch health care to new future?
- Can we hack our way to better health care?
Big data, big pharma and you. All of you.
by Tom Ulrich on October 20, 2014
 Where is the next generation of therapeutic innovations going to come from? Population-level genomic studies? The fitness trackers on everyone’s wrist? Mining electronic medical records? People’s tweets, Yelps and Facebook posts?
Where is the next generation of therapeutic innovations going to come from? Population-level genomic studies? The fitness trackers on everyone’s wrist? Mining electronic medical records? People’s tweets, Yelps and Facebook posts?
How about all of the above?
What all of these things have in common is data. Lots of it. Some of it represents kinds of data that didn’t exist 5 or 10 years ago, but all of it is slowly beginning to fuel the pharma sector’s efforts to create the next blockbuster drug or targeted therapeutic.
At least, it should be. Full story »
The sharks are circling health care
by Nancy Fliesler on October 17, 2014

Health care institutions, universities and even the U.S. government are helping innovators hone their pitches and get backing.
It’s a great infotainment formula—even my 10-year-old daughter is a fan—but it’s also a hit beyond the living room. Health care organizations are increasingly borrowing the “Shark Tank” script to get new ideas or to bankroll their own innovations. Boston Children’s Hospital is doing so at our Global Pediatric Innovation Summit + Awards (Oct. 30-31), bringing in “Shark Tank”’s Daymond John to moderate. But we’re certainly not alone. In recent months: Full story »
Related posts:
Leveraging bacteria biofilms for vaccines
by Alice McCarthy on October 14, 2014
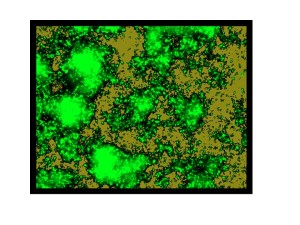
Through genetic engineering, this Vibrio cholerae biofilm can be loaded with extra antigens, creating a super-charged but inexpensive vaccine.
Paula Watnick, MD, PhD, an infectious disease specialist at Boston Children’s Hospital, has a plan that stems from her work on cholera: using a substance produced by the bacteria themselves to make inexpensive and better vaccines against them.
Cells do all the work
Bacteria produce biofilms—a sticky, tough material composed of proteins, DNA and sugars—to help them attach to surfaces and survive. Full story »
Related posts:
Caution: Combustion ahead at Innovation Summit
by Ivan Salgo on October 9, 2014
 Ivan Salgo, MD, MBA, is Senior Director, Cardiology, Philips Ultrasound.
Ivan Salgo, MD, MBA, is Senior Director, Cardiology, Philips Ultrasound.
I’m thrilled to be part of the judging panel for the Innovation Tank at Boston Children’s Hospital’s upcoming Global Pediatric Innovation Summit + Awards. I can’t imagine a better way to bring together a duly combustible mixture of new thoughts and ideas with the spark of innovation.
It’s very clear that the pace of innovation needs to accelerate.
The Innovation Tank provides a rich and engaging way to accelerate innovation by putting the best ideas and technologies in front of people who can incubate and fund them, and ultimately, take them to success.
The genius of the Innovation Tank is that it coalesces a critical mass of people and ideas in a single place. By bringing ideas to the front through competition, participants can raise the bar on their own creativity to bring forth compelling ideas that matter to medicine, that matter to peoples’ lives. Full story »
The backstory behind organs-on-chips
by Tom Ulrich on October 8, 2014
With the launch this summer of Emulate Inc., organs-on-chips—a disease-modeling platform we’ve covered several times on Vector—made the jump from academic to commercial development.
Though developed at the Wyss Institute for Biologically Inspired Engineering, the chips’ story actually began more than 20 years ago in Boston Children’s Hospital’s Vascular Biology Program (VBP). It’s a story that brings together characters from multiple fields and emerges from one fundamental concept: that mechanical forces are critical to the function and fate of cells, tissues and organs. Full story »
Innovation inspiration: From Shakira to Toys ‘R’ Us
by Lisa Fratt on October 7, 2014

Start your engines: A fleet of GoBabyGo cars, customized by therapists and parents to give disabled children mobility and help strengthen weak muscles. (Courtesy Cole Galloway)
$100 plastic car stands in for $25,000 power wheelchair
In the first (and likely only) National Institutes of Health-funded shopping spree at Toys R’ Us, Cole Galloway, director of the Pediatric Mobility Lab at the University of Delaware, and crew stocked up on pint-sized riding toys.
Galloway’s quest was to facilitate independence and mobility among disabled children from the age of six months and older and offer a low-tech solution during the five-year wait in the United States for a $25,000 power pediatric wheelchair.
The hackers jerry-rigged the toys with pool noodles, PVC pipe and switches, reconfiguring them as mobile rehabilitation devices to promote functional skills among kids with special needs. Full story »
Related posts:
- Innovation Day at Children’s Hospital Boston: A preview
- Top 10 science and clinical innovation trends: Looking forward to 2012
- OPENPediatrics: A blueprint for big innovation
- “Intellectuals solve problems, geniuses prevent them”
- Serendipity in science: Collaborating to build robotic clothing for brain-damaged children
Four attributes of an innovation culture
by Naomi Fried on October 2, 2014
 Naomi Fried, PhD, Chief Innovation Officer at Boston Children’s Hospital, will lead a panel on Innovation Acceleration at Taking on Tomorrow: Global Pediatric Innovation Summit + Awards (October 30-31, Seaport World Trade Center, Boston). Register now!
Naomi Fried, PhD, Chief Innovation Officer at Boston Children’s Hospital, will lead a panel on Innovation Acceleration at Taking on Tomorrow: Global Pediatric Innovation Summit + Awards (October 30-31, Seaport World Trade Center, Boston). Register now!
The word innovation gets thrown around a lot these days by people trying to set their products and ideas apart in the marketplace. But when everything is innovative, is anything really innovative? And if there really are innovative ideas, are they simply flashes of brilliance that can’t be planned for or predicted?
The answer to this last question is “no,” as I see every day at Boston Children’s Hospital, where I lead the Innovation Acceleration Program. The real trick is creating an innovation culture that supports great ideas—but that also supports the not-so-great ideas that teach us almost as much.
So what are the attributes of an innovation culture? Full story »