Where is the next generation of therapeutic innovations going to come from? Population-level genomic studies? The fitness trackers on everyone’s wrist? Mining electronic medical records? People’s tweets, Yelps and Facebook posts?
Where is the next generation of therapeutic innovations going to come from? Population-level genomic studies? The fitness trackers on everyone’s wrist? Mining electronic medical records? People’s tweets, Yelps and Facebook posts?
How about all of the above?
What all of these things have in common is data. Lots of it. Some of it represents kinds of data that didn’t exist 5 or 10 years ago, but all of it is slowly beginning to fuel the pharma sector’s efforts to create the next blockbuster drug or targeted therapeutic.
At least, it should be. Full story »
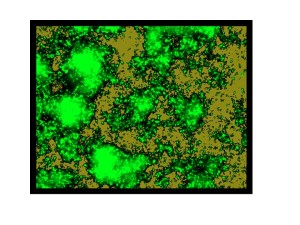
Through genetic engineering, this Vibrio cholerae biofilm can be loaded with extra antigens, creating a super-charged but inexpensive vaccine.
Malaria. Cholera. Now Ebola. Whatever the contagion, the need for new, or better, vaccines is a constant. For some of the most devastating public health epidemics, which often break out in resource-poor countries, vaccines have to be not only medically effective but also inexpensive. That means easy to produce, store and deliver.
Paula Watnick, MD, PhD, an infectious disease specialist at Boston Children’s Hospital, has a plan that stems from her work on cholera: using a substance produced by the bacteria themselves to make inexpensive and better vaccines against them.
Cells do all the work
Bacteria produce biofilms—a sticky, tough material composed of proteins, DNA and sugars—to help them attach to surfaces and survive. Full story »
(gegenart/Shutterstock)
A mouse surrounded by computer screens turns its head when it notices lines moving across one of them, as a camera captures this evidence of visual acuity. A chamber similarly equipped with video cameras tests social interaction between mice. A small swimming pool, with shapes on its walls as navigational cues, lets scientists gauge a mouse’s spatial memory. A pint-sized treadmill, with a tiny camera to watch foot placement, measures gait.
Here in the Neurobehavioral Developmental Core at Boston Children’s Hospital, managed by Nick Andrews, PhD, the well-tended mice also have opportunities to play: “If you have a happy mouse,” says Andrews, “researchers get better, more consistent results.” Full story »
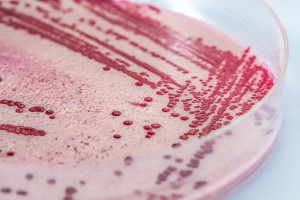
Listeria bacteria on a plate. The biology of HIV/AIDS suggests T-cells have a hitherto unrecognized way of killing pathogens like these.
The immune system, despite its immense complexity, really has only a few ways to kill bacteria:
- Neutrophils and macrophages can capture and digest extracellular bacteria (ones that live free in tissues and the bloodstream).
- Peptides (protein fragments) can punch holes in bacterial membranes or cross the membranes to disrupt bacterial processes.
- T-cells can kill cells infected by intracellular bacteria (ones that take up residence within cells).
It’s this last mechanism that I want you to pay attention to. The conventional wisdom has long held that T-cells can only kill intracellular bacteria indirectly by eliminating the cells they’ve infected. But a paper by Judy Lieberman, MD, PhD, of Boston Children’s Hospital’s Program in Cellular and Molecular Medicine, reveals that T-cells have a hitherto unnoticed way of directly killing intracellular bacteria And she only found it because of HIV/AIDS. Full story »

Academic and industry partners are explicitly working to fill pharma pipelines.
Academic researchers and physician innovators are great at making research discoveries and developing inventions at an early stage. But if you were to fund them to turn their research findings into a product, would they have the expertise and experience needed to be successful? Most would not.
The investment community talks about the innovation funding gap, a.k.a. the “valley of death.” But there is also a knowledge gap on the part of academic researchers when it comes to transforming their technologies into therapeutics. Most want their findings to lead to new treatments for patients, but they lack the experience and expertise that companies have to advance early-stage research to a clinical stage. That includes expertise in designing pre-clinical experiments and navigating regulatory pathways for commercial development.
Academics often enter agreements with pharmaceutical companies, many of which are early-stage research grants. Often, these industry-sponsored research projects end with a scientific publication and are unsuccessful in generating new therapeutics—a subpar outcome for the company investor. Full story »
What's drawing lung cancer cells to migrate? (Juan Gaertner/Shutterstock)
Ninety percent of lung cancer deaths are caused by the tumor’s spread—or metastasis—to other organs. Researchers have now discovered an approach to blocking metastasis in the most common type of lung cancer, adenocarcinoma, that potentially could be added to chemotherapy treatments aimed at shrinking the primary tumor.
Kerstin Sinkevicius, PhD, a research fellow at Boston Children’s Hospital, started with this question: Is there anything in a lung tumor’s environment that makes it metastasize? She sampled tissue from human lymph nodes—the first place cancers typically spread to—to see if the cells there were secreting anything that might lure cancer cells to migrate.
One chemical stood out: a growth factor called brain-derived neurotrophic factor, or BDNF. Secreted near maturing neurons, BDNF is best known for its role in stimulating the developing nervous system. Full story »
Mast cells don’t simply cause acute allergic reactions. They also turn off immune tolerance. But that could change. (Bruce Blaus/Wikimedia Commons)
Hans Oettgen, MD, PhD, is Associate Chief of the Division of Allergy and Immunology at Boston Children’s Hospital. He leads a research group investigating mechanisms of allergic diseases.
Not long ago I received a wonderful email from “Sam,” an 18-year-old young man with peanut allergy. He was participating in a clinical trial of oral immunotherapy (OIT) being carried out by colleagues here at Boston Children’s Hospital.
In OIT, patients receive initially minute doses of the food to which they are allergic. Then, over many weeks, they ingest increasing amounts, under close medical monitoring at the hospital.
OIT’s goal is to get patients to tolerate previously allergenic foods by inducing their bodies to produce Treg cells, or regulatory T cells. These are the master controllers of our immune responses, and their actions include suppressing allergic responses to foods. Food ingestion, as in OIT, will eventually induce food-specific Treg cells, but it can be a long and cumbersome process. For Sam, ingesting escalating doses of peanuts proved difficult: His email described frequent reactions ranging from stomachaches and itchiness to difficulty breathing. Full story »

A new proposal suggests spreading drug development risk among many small investors.
Ed Anderson, CCRP, is a clinical research specialist for the Clinical Research Center’s Development and Operations Core at Boston Children’s Hospital.
There’s no way around it. Obtaining approval to market a new drug is lengthy, complex, costly and fraught with uncertainty and risk. Financial engineers at MIT propose a strategy to minimize that risk—one that deserves a close look.
In the last 10 years, the aggregate cost of pharmaceutical research and development has doubled, but the number of approved products has remained the same. To compound the problem, a $1.6 billion reduction in NIH funding, caused by the 2013 sequester, has stalled research projects at more than 2,500 research institutions supported by grants. Pressure from investors and stakeholders is pushing pharmaceutical companies to focus on projects with a greater chance of financial success.
As a result, translational studies—those that bridge the gap between basic research and clinical trials—continue to be neglected and account for less than 12 percent of total research funding. Full story »
 You’d think drugs meant to be taken by children for years would be studied in children for a long time to measure their long-term safety.
You’d think drugs meant to be taken by children for years would be studied in children for a long time to measure their long-term safety.
You’d think drugs for a condition affecting millions of children would be tested in many, many children to catch any rare side effects.
You’d think all this would happen before the Food and Drug Administration, an agency known for its strict criteria, approved them for marketing.
But if a new PLoS ONE paper by Boston Children’s Hospital’s Florence Bourgeois, MD, MPH, and Kenneth Mandl, MD, MPH, is any indication, you’d be wrong.
In it, the pair reports that the FDA approved 20 attention deficit hyperactivity disorder (ADHD) drugs over the last 60 years without what would be considered sufficient long-term safety and rare adverse event data.
Their findings, they say, point to larger issues in how the FDA’s approval process addresses the long-term safety of drugs intended for chronic use in children. Full story »
 Where is the next generation of therapeutic innovations going to come from? Population-level genomic studies? The fitness trackers on everyone’s wrist? Mining electronic medical records? People’s tweets, Yelps and Facebook posts?
Where is the next generation of therapeutic innovations going to come from? Population-level genomic studies? The fitness trackers on everyone’s wrist? Mining electronic medical records? People’s tweets, Yelps and Facebook posts?