Parents, clinicians, app developers, designers and more had 18 hours to prototype digital healthcare solutions at Hacking Pediatrics, produced by Boston Children’s Hospital and MIT Hacking Medicine. To accompany our earlier post, we created this Storify. Full story »
Posts by author:
Nancy Fliesler
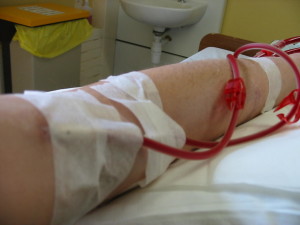
Understanding the genetic causes of nephrotic syndrome could lead to better drug treatments that reduce the need for dialysis or a kidney transplant. (Image: Wikimedia Commons)
Until recently, no one knew what caused nephrotic syndrome; the first causative gene was identified just a dozen years ago. The lab of Friedhelm Hildebrandt, MD, PhD, at Boston Children’s Hospital is one of a handful that’s been chipping away at the others.
Hildebrandt receives, on average, one blood sample a day from patients all over the world. Full story »
“There are two kinds of somatic mutations that get missed,” says Christopher Walsh, MD, PhD, chief of Genetics and Genomics at Boston Children’s Hospital. “One is mutations that are limited to specific tissues: If we do a blood test, but the mutation is only in the brain, we won’t find it. Other mutations may be in all tissues but in only a fraction of the cells—a mosaic pattern. These could be detectable through a blood test in the clinic but aren’t common enough to be easily detectable.”
That’s where deep sequencing comes in. Reporting last month in The New England Journal of Medicine, Walsh and postdoctoral fellow Saumya Jamuar, MD, used the technique in 158 patients with brain malformations of unknown genetic cause, some from Walsh’s clinic, who had symptoms such as seizures, intellectual disability and speech and language impairments. Full story »
Going back to World War II, it’s been speculated that recovery of peripheral nerve injuries—like those in limbs and extremities—is influenced by age. And studies indicate that peripheral neuropathy is common in people over 65, including those who have received cancer chemotherapy, and often unexplained.
“When you’re very young, the system is very plastic and able to regenerate,” Michio Painter told me recently. He is a graduate student in the laboratory of Clifford Woolf, PhD, director of the F.M. Kirby Neurobiology Center at Boston Children’s Hospital. “After that, there’s a gradual decline. By the age of 30, much of this plasticity is gone.”
Traditionally, this decline has been thought to reflect age-related differences in neurons’ ability to regrow, but when Painter studied neurons in a dish, he couldn’t confirm this. Full story »
As in this map on the cover of Cell, a cell has many possible destinations or “fates,” and can arrive at them through three main stem cell engineering methods:
• reprogramming (dialing a specialized cell, such as a skin cell, back to a stem-like state with full tissue-making potential)
• differentiation (pushing a stem cell to become a particular cell type, such as a blood cell)
• direct conversion (changing one kind of specialized cell to another kind)
Freely available on the Internet, CellNet provides clues to which methods of cellular engineering are most effective—and acts as a much-needed quality control tool. Full story »

Emmie Mendes was lucky enough to be diagnosed before age 3, but many families face a much longer journey.
At first, Corrie and Adam Mendes thought their daughter Emmie had an inner ear problem. She was late with several early milestones, including walking, and when she did walk, she often lost her balance. The family pediatrician sent them to a neurologist, who ordered a brain MRI and diagnosed her with pachygyria, a rare condition in which the brain is smoother than normal, lacking its usual number of folds.
Additionally, Emmie’s ventricles, the fluid-filled cushions around the brain, looked enlarged, so the neurologist recommended brain surgery to install a shunt to drain off fluid. He advised Corrie and Adam that Emmie’s life expectancy would be greatly reduced.
As Corrie recounts on her blog, Emmie’s Story, she went online and came across the research laboratory of Christopher Walsh, MD, PhD, at Boston Children’s Hospital. The lab does research on brain malformations and has an affiliated Brain Development and Genetics Clinic that can provide medical care.
After Walsh’s team reviewed Emmie’s MRI scan, genetic counselor Brenda Barry invited the family up from Florida. Full story »
At TEDx Longwood this spring, Leonard Zon, MD, founder and director of the Stem Cell Program at Boston Children’s Hospital, took the stage. In his enthusiastic yet humble style, he took the audience on a journey that included time-lapse video of zebrafish embryos developing, a riff by Jay Leno and a comparison of stem cell “engraftment” to a college kid coming home after finals: “You sleep for three days, and on day 4, you wake up and you’re in your own bed.” Three takeaways:
1) Stem cells made from our own skin cells can help find new therapeutics. With the right handling, they themselves can be therapeutics, producing healthy muscle, insulin-secreting cells, pretty much anything we need. (So far, this has just been done in mice.)
2) Zebrafish, especially when they’re see-through, can teach us how stem cells work and can be used for mass screening of potential drugs. The Zon Lab boasts 300,000 of these aquarium fish, and can mount robust “clinical trials” with 100 fish per group.
3) Drugs discovered via zebrafish are in human clinical trials right now: A drug to enhance cord blood transplants for leukemia or lymphoma, and an anti-melanoma drug originally used to treat arthritis.
Zon, who co-founded the biopharm company Fate Therapeutics, will be part of a judging panel of clinicians and venture capitalists for the Innovation Tank at Boston Children’s Global Pediatric Innovation Summit + Awards (Oct. 30-31). Don’t miss it!