People who have had a heart attack or have coronary artery disease often sustain damage that weakens their heart. Milder forms of heart failure can be treated with medications, but advanced heart dysfunction requires surgery or heart transplant. A team of physicians, engineers and materials scientists at Children’s Hospital Boston and MIT offers two alternative ways to strengthen weakened, scarred heart tissue — both involving nanotechnology.
One approach blends nanotechnology with tissue engineering to create a heart patch laced with gold whose cells all beat in time – as shown in the above video.
The other uses minute nanoparticles that can find their way to dying heart tissue, carrying stem cells, growth factors, drugs and other therapeutic compounds.
A heart of gold
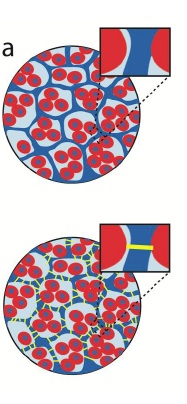
Engineered heart tissue with (bottom) and without gold nanowires. Cardiac cells are in red, scaffolding material in blue and nanowires in yellow.
Tissue-engineered cardiac patches are starting to go into clinical trials for heart patients. They’re made by seeding heart cells onto porous scaffolds that give the tissue shape and organization. But there’s one problem: The heart is an electrically conductive organ, and the scaffolding used for the patches isn’t conductive, so the tissue doesn’t contract as normal heart tissue does.
Tal Dvir and Brian Timko, postdocs in both the Laboratory for Biomaterials and Drug Delivery at Children’s, headed by Daniel Kohane, and the lab of Robert Langer at MIT - came up with an idea: sprinkling tiny gold wires into the patches to enhance their electrical conductivity.

The gold nanowires average 30 nanometers thick and 2-3 microns long, just barely visible to the naked eye. Gold is easy to fabricate and well tolerated by the body.
The gold-laced patches, described in Nature Nanotechnology, were thicker and their cells better organized. They had ramped-up production of proteins involved in muscle calcium binding and contraction and electrical coupling between cells. And, when stimulated with an electrical current, the cells produced a measurable spike in voltage.
The nanowires seem to work by poking through the scaffolding material, which normally impedes electrical conduction, enhancing communication between adjacent groups of heart cells. Without the wires, patches produced a negligible current, and cells beat only in small clusters right at the source of stimulation.
“With the nanowires, you see a lot of cells contracting together, even when the stimulation is far away,” says Kohane. “That shows the tissue is conducting.”
Kohane’s team hopes to test the gold-wired patches in live animal models soon.
Guided missiles
But he and Dvir also have their eye on another approach, just reported in Nano Letters. It uses nanotechnology to create tiny guided missiles that can be injected intravenously, circulate in the blood, then exit at the heart and target tissue damaged by heart attack.
Many current experimental approaches to heart attack involve supplying growth factors, drugs, stem cells and other therapeutic agents to the scarred, dying tissue. Some of these compounds, such as periostin and neuregulin, have been shown in animal models to enhance heart regeneration and improve cardiac function. But the existing delivery approaches are all invasive, involving direct injections into the heart, catheter procedures, or surgical placement of implants that release the necessary factors.
Envisioning a simpler, gentler approach, Kohane, Dvir and colleagues fabricated liposomes – microscopic fat-coated spheres that can be filled with therapeutic compounds. To their surfaces they attached a molecule that, similar to an antibody, recognizes a specific protein – in this case, one made in large amounts after heart attacks, known as angiotensin II type 1 receptor.
Studying mice with induced heart attacks, with critical help from Ronglih Liao and Michael Bauer at Brigham and Women’s Hospital, they show that the liposomes, injected into a vein, were small enough to slip through leaky blood vessels near the infarcted heart and bind to the scarred heart tissue.
“If we inject these particles in healthy mice, they don’t go to the heart but continue circulating in the body,” says Dvir, now at Tel Aviv University. “For the first time, we’ve shown that nanoparticles can circulate in the bloodstream and target the heart, specifically after a heart attack. In principle, we can bring drugs, growth factors, and do many things specifically in the injured area.”
The ultimate goal is to have the particles release compounds that promote regeneration. One approach is to release factors that attract the patient’s own stem cells, avoiding the need for tissue-engineered patches. But to date, no one’s gotten stem cells to differentiate efficiently into cardiomyocytes (heart muscle cells), Dvir says. Still, it’s possible the stem cells could differentiate to blood-vessel-forming cells that would also be of benefit after heart attack.
“For now, bringing a living patch with contracting cells that could integrate into the healthy part of the heart is the way do it,” says Dvir. “I hope that in five to ten years, both of these approaches could be helpful in the clinic.”
Ed. note: Read Dvir’s blog post about combining tissue engineering with nanotechnology, and our recent post on using liposomes to block HIV infection.