Measuring the total amount of DNA damage within a tumor’s cells could help doctors predict its vulnerability to drugs like cisplatin. (Haukeland universitetssjukehus/Flickr)
Drugs like cisplatin that break DNA are some of the strongest weapons we have against breast, ovarian and other cancers. The problem, common to every form of chemotherapy, is that cisplatin doesn’t work for everyone. Given the potential side effects that go along with the drug—including vomiting, hearing loss and muscle cramps, just to name a few—the decision to give it to a patient becomes something of a gamble: Does the benefit outweigh the risk?
There are tests that examine individual genes and which can give doctors a limited view as to which tumors might respond best to cisplatin. But a multicenter team co-led by Zoltan Szallasi, MD, of Boston Children’s Hospital’s Informatics Program (CHIP), thinks they may have a solution that looks beyond individual genes to see which tumors might succumb to cisplatin and other drugs like it. Full story »
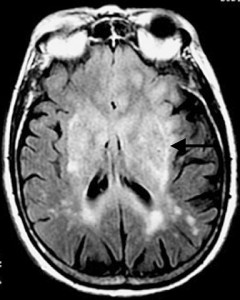
Brain tumors like the diffuse, light gray one in this MRI do a remarkably good job of hiding from the immune system. A new treatment based on gene therapy could strip their camouflage away. (Filip Em/Wikimedia Commons)
If there’s anything that tumors are good at, it’s hiding themselves. Not from things like MRIs or CT scans, mind you, but from the immune system. Since a tumor grows from what were at one time normal, healthy cells it’s still “self,” still one of the tissues that makes you you.
“Tumor cells display very subtle differences that distinguish them from healthy cells, but by and large they look the same to your immune system,” says Mark Kieran, a pediatric neuro-oncologist at the Dana-Farber/Children’s Hospital Cancer Center and Children’s Hospital’s Vascular Biology Program. “The question is: How can we unmask tumors so that the immune system can do its job?”
Researchers have worked for years on cancer vaccines aimed at getting the immune system to wake up to the presence of a tumor and turn on it. With a Phase 1 safety trial , Kieran and his colleagues, including Children’s neurosurgical oncologist Lily Goumnerova, are evaluating a different strategy for patients with hard-to-treat brain tumors called malignant gliomas: They’re giving the tumors a cold. Full story »

Cancer stem cells’ clever defenses may be the seeds of their undoing. (Image: leftover bacon/OpenClipArt)
Some scientists still debate the existence of cancer stem cells – rare cells that can singlehandedly perpetuate a tumor, and possibly make it more aggressive. But others have moved on, isolating candidate cancer stem cells and documenting their distinctive characteristics and markers.
And some are starting to figure out how these cells operate and leverage that knowledge to come up with new approaches to cancer therapy.
Children’s scientist Markus Frank has been building quite a dossier on cancer stem cells, starting with melanoma stem cells. “Many of the features that make a cancer bad seem to be localized in this subpopulation of cells,” he says. Full story »
Image by Steve Case via TwitPic: http://twitpic.com/31dosf
Before Children’s Hospital Boston’s own Frances Jensen, Director of Epilepsy Research, took the stage yesterday, Richard Saul Wurman, organizer of TEDMED and the TED conferences, spoke warmly of Children’s participation and sponsorship of this year’s event. A generous gift from the Hassenfeld Family Initiatives enabled that participation, and Wurman thanked the Hassenfelds and Children’s for bringing “such interesting people” to TEDMED 2010. With that, Jensen began her talk about the importance of understanding the developing brain. Full story »










