(David Sky, Creative Commons License 2004-2011)
“Value is more important than innovation,” declared Angus Russell, CEO of Shire Pharmaceuticals, in his opening keynote address at Biopharm America last week. At a drug development conference, where attendees typically focus on interesting new ways to address therapeutic problems, that sounded a bit heretical.
But it was a telling example of how cost pressures are now manifesting in pharma. Some complex treatments can be new and innovative, but not provide measurable improvement in patient outcomes. Can we conclude that they have no value? This has been one of the central questions fueling the debate over U.S. healthcare reform, and will influence development of medicines into the future.
Hospitals and others have been asked for some time to show that they can provide the same level of care for lower cost. Now, it’s pharma’s turn. Full story »
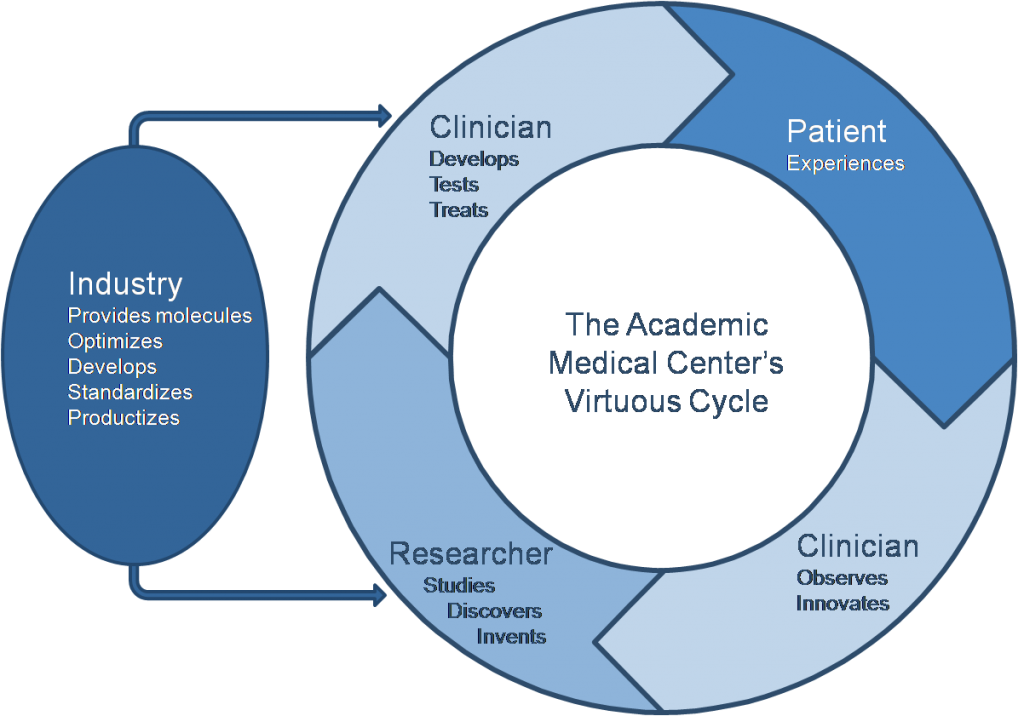
The virtuous cycle of treatment development, and how it can be enhanced by partnership with industry.
When the clinical and research enterprises at an academic medical center are strategically and tactically aligned, they enter a “virtuous cycle,” described by Stephen Wartman, president of the Association of Academic Health Centers. Clinician-researchers embody it: They know their patients’ needs, what treatments are available and the frustration when those options are insufficient. They take clinical problems back to the lab to figure out solutions, and are highly motivated to develop products because their goal begins and ends with helping the patient.
The academic medical center is the epicenter of therapeutic development. It’s where potential drug targets are identified and mapped from animal models to human tissues, and where clinical trials will ultimately take place. Yet, in the past, pharmaceutical and biotech companies have driven drug development, while academia has been a piecemeal contributor Full story »

(photo: kisforkate/Flickr)
In almost every conversation I have these days with potential industry partners, I hear what seems to be the new buzz phrase: “Clear path to clinic.” A molecule with a “clear path to clinic” can become a medicine quickly, by virtue of a well-defined patient population, clear endpoints for clinical trials and measurable biomarkers.
Industry is increasingly recognizing that investigators at academic research centers know this path to clinic best. This has brought a shift in how industry interacts with academia, and new and equitable partnership structures are poised to facilitate joint therapeutic development like never before.
Pharma companies have a strong incentive to change. Full story »
 Over the past nine months, Pfizer has built collaborations with a number of premiere academic medical centers, including Children’s Hospital Boston. Wednesday marked the launch of the Boston branch of Pfizer’s Centers for Therapeutic Innovation (CTI), fostering independent collaborations with seven Boston institutions. The CTI aims to facilitate and support joint drug discovery and development — from the conception of an idea through early clinical trials.
Over the past nine months, Pfizer has built collaborations with a number of premiere academic medical centers, including Children’s Hospital Boston. Wednesday marked the launch of the Boston branch of Pfizer’s Centers for Therapeutic Innovation (CTI), fostering independent collaborations with seven Boston institutions. The CTI aims to facilitate and support joint drug discovery and development — from the conception of an idea through early clinical trials.
So why is Children’s Hospital Boston, the #1 pediatric hospital in the country with an annual research base of $225 million, entering into a partnership with Pfizer? Simply, Pfizer has complementary knowledge, resources and infrastructure to support a number of our therapeutic projects. Pfizer, and other companies, can help us move early-stage discoveries out of the lab and safely into the clinic more quickly than we could on our own, ultimately supporting our mission. Full story »
 You’ll almost never hear innovators say, ‘Can we do something?’ You will sometimes hear them say, ‘How can we do something?’
You’ll almost never hear innovators say, ‘Can we do something?’ You will sometimes hear them say, ‘How can we do something?’
I heard this last week from Kim Smith, a founding team member of Teach For America and founder/CEO of Bellwether Education Partners, at MIT’s Innovation in Healthcare Symposium. It reminded me of the innovators at our hospital, whose problem-solving visions we try to push toward real products.
Solving problems in the health care system itself seemed a far more daunting task. I arrived at the symposium thinking about the entrenched interests keeping current systems in place — the way doctors are trained, the way companies in health care create competitive barriers to information sharing, the pharmaceutical industry’s business model, the fact that insurance companies are incentivized not to cover sick people. The list goes on.
But I left this gathering feeling uplifted and inspired. Full story »
 Developing treatments for a debilitating disorder for which there is no existing therapy, with a genetic component and early onset in childhood that affects thousands of people in the world, but not millions- this sounds like what Children’s Hospital Boston investigators do every day, but it may not sound like a compelling business opportunity for big companies who have made their net worth by selling large volumes of pills to millions of people. But the trend is clear- Full story »
Developing treatments for a debilitating disorder for which there is no existing therapy, with a genetic component and early onset in childhood that affects thousands of people in the world, but not millions- this sounds like what Children’s Hospital Boston investigators do every day, but it may not sound like a compelling business opportunity for big companies who have made their net worth by selling large volumes of pills to millions of people. But the trend is clear- Full story »
 TEDMED was fascinating, and it was a great experience for the Children’s team who attended. Based on the conference attendees and the intimate size of the gathering, it offered our constituents ample opportunity to interact with peers from other fields, policy decision makers, media influencers, future collaborators and potential sponsors in a way that wouldn’t have been possible in any other setting. Full story »
TEDMED was fascinating, and it was a great experience for the Children’s team who attended. Based on the conference attendees and the intimate size of the gathering, it offered our constituents ample opportunity to interact with peers from other fields, policy decision makers, media influencers, future collaborators and potential sponsors in a way that wouldn’t have been possible in any other setting. Full story »
 This is how Dean Kamen, the consummate inventor, framed his talk on Day 3 at TEDMED. If this is the definition of genius, then Marc Koska, who developed a syringe which cannot be reused and also spoke yesterday, certainly is one. The syringe and the associated public health campaign he ran in India and other parts of the developing world to raise awareness about the danger of reusing needles, common practice in these countries, is credited with saving 10 million lives.
This is how Dean Kamen, the consummate inventor, framed his talk on Day 3 at TEDMED. If this is the definition of genius, then Marc Koska, who developed a syringe which cannot be reused and also spoke yesterday, certainly is one. The syringe and the associated public health campaign he ran in India and other parts of the developing world to raise awareness about the danger of reusing needles, common practice in these countries, is credited with saving 10 million lives.
Both Kamen’s and Koska’s presentations touched on a theme that hasn’t been discussed much here: the interface between the technology inventor and the private sector industry that develops, manufactures, markets and distributes products. Full story »
Image by Steve Case via TwitPic: http://twitpic.com/31dosf
Before Children’s Hospital Boston’s own Frances Jensen, Director of Epilepsy Research, took the stage yesterday, Richard Saul Wurman, organizer of TEDMED and the TED conferences, spoke warmly of Children’s participation and sponsorship of this year’s event. A generous gift from the Hassenfeld Family Initiatives enabled that participation, and Wurman thanked the Hassenfelds and Children’s for bringing “such interesting people” to TEDMED 2010. With that, Jensen began her talk about the importance of understanding the developing brain. Full story »
Yes, it’s true. If you were following on Twitter, you would have seen the tweets about Martha Stewart at the TEDMED conference touching a pair of breathing pig lungs on stage. The setup was a miraculous opening aria by opera singer Charity Tilleman-Dick, who then revealed the real miracle: a year ago she had a double-lung transplant. Shaf Keshavjee followed, wheeling out his ex vivo lung machine, which can maintain a healthy lung out of the body for 24 hours, enabling doctors to treat the lung with medications and even gene therapy before transplanting it to enhance the chances of success. Keshavjee invited audience members to come and touch the lungs, which is how Martha ended up there, iPhone in hand, preparing to tweet about her experience. Full story »















