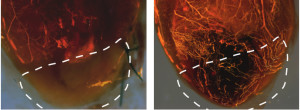
In mice, VEGF-A modRNA visibly improved blood supply to heart muscle (right image) as compared with no treatment.
Researchers have struggled to find the right approach to regeneration. Cell transplants have been tried, but the cells don’t engraft well long term and haven’t shown efficacy. Gene therapy to spur regeneration has been tested in animals, but dosage is hard to control and there’s a risk of genes going where they shouldn’t, causing tumors and other problems. Protein drugs have been tried, but they have short half-lives, being degraded or eliminated by the body before they can do much good. They are also hard to target to the heart.
A more recent approach to cardiac regeneration is to stimulate the body itself—and, specifically, progenitor cells— to repair the heart from within. One method tested recently uses microRNAs—molecules that regulate gene activity—to multiply cardiomyocytes. And in September, reporting in Nature Biotechnology, a team of researchers at Boston Children’s Hospital, Massachusetts General Hospital and the Harvard Stem Cell Institute (HSCI) demonstrated another tactic: Turning on the heart’s own cellular growth signals with modified messenger RNAs.
The modified RNA approach was first developed by Derrick Rossi, PhD, of the HSCI and Boston Children’s Program in Cellular and Molecular Medicine. Rossi took the messenger RNAs that provide genetic blueprints for making proteins and chemically tweaked them to avoid triggering an immune response (see the video below for a tutorial).
Acts like a drug, works like a gene
In March 2013, the startup company that grew out of this technology, Moderna, entered a $240 million agreement with AstraZeneca to develop a variety of modRNA drugs, which spur the body itself to make what it needs.
“The modified RNA approach combines the good parts of both drug therapy and gene therapy,” says William Pu, MD, an HSCI member and a cardiologist at Boston Children’s, and a co-corresponding author on the Nature Biotechnology paper. “Just like a drug, modified RNA acts for a time and then goes away. And because it’s a gene, you know what it does, and you don’t have to develop a drug that substitutes for it.”
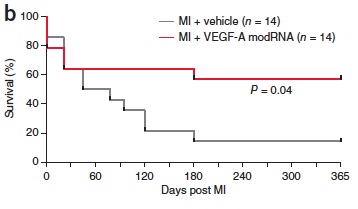
Senior study investigator Kenneth Chien, MD, PhD, of HSCI and Massachusetts General Hospital (now at the Karolinksa Institutet), envisioned modRNAs as a way to get injured heart tissue to regenerate. With Lior Zangi and Kathy Lui in his lab, he showed that a modified RNA encoding vascular endothelial growth factor (VEGF), injected near the infarct in a mouse model of heart attack, not only improved heart function (left-ventricular function in particular) but also increased one-year survival from under 20 percent to just under 60 percent:
Zangi then worked with the Pu lab to zero in on the mechanism. They showed that the VEGF-A modRNA worked primarily by pushing progenitor cells in the heart to grow and become endothelial cells that make blood vessels. While VEGF is known to promote expansion of existing blood vessels, this action of changing progenitors’ fate was unexpected. The modRNA also pushed the progenitor cells to form cardiomyocytes, although this effect was more modest.
And, importantly, the modRNA efficiently penetrated heart muscle with minimal toxicity. Even a single dose, simulating natural signals from cells, was enough to improve myocardial outcome and survival. “If you give a high, transient pulse of a paracrine signal, you can push a progenitor cell to do something therapeutically beneficial, and have a sustained effect,” says Pu. “We’re attracted to signaling molecules because that’s how nature does it.”
Timing is everything
VEGF has been studied for almost two decades as a way of helping people recover from heart attacks by encouraging growth of collateral vessels. But results have been mixed in animal models and poor in human studies.
“We think that’s because of the delivery technique,” says Pu. “We found that the amount and timing really matter.”
In mice, the VEGF-A modRNA treatment only worked in the context of injury, so it would likely need to be given fairly soon after a heart attack—right when the body is trying to rally its own response. And a quick, brief pulse seems to work best: Long-term expression of VEGF (delivered by a gene) actually reduced mouse survival after myocardial infarction, or heart attack, leading to production of dysfunctional, leaky vessels, Pu says.
Here’s a recap of how modRNA works—as originally applied in stem cell research:







