In 1982, insulin became the first FDA-approved protein drug created through recombinant DNA technology. It was made by inserting the human insulin gene into a bacterial cell’s DNA, multiplying the bacteria and capturing and purifying the human insulin in bioreactors. Full story »
Posts tagged as:
tissue engineering
 Scaffolds made of silk could give doctors a simple, more effective material for performing bladder augmentation in people with urinary tract defects—to relieve incontinence and prevent kidney damage in children born with small bladders, for example. Rather than using cells to augment the bladder, a complicated process, silk could provide an “off the shelf” option, says Carlos Estrada, MD, a urologist at Boston Children’s Hospital.
Scaffolds made of silk could give doctors a simple, more effective material for performing bladder augmentation in people with urinary tract defects—to relieve incontinence and prevent kidney damage in children born with small bladders, for example. Rather than using cells to augment the bladder, a complicated process, silk could provide an “off the shelf” option, says Carlos Estrada, MD, a urologist at Boston Children’s Hospital.
Recent research by Estrada and Joshua Mauney, PhD, shows that scaffolds made of fibroin (the protein that makes up raw silk) have worked well in augmenting bladders in animal models—without the need for cells.
Estrada and Mauney built on the work of Anthony Atala, MD, who became head of the Institute for Regenerative Medicine at Wake Forest after undertaking pioneer work in tissue engineering in Boston Children’s Urology Department. Full story »
The anterior cruciate ligament (ACL) is a powerhouse and the perplexing nexus of a sports injury epidemic.
Providing primary stability across the knee joint, the ACL is remarkably susceptible to rupture or tear, with more than 400,000 surgical reconstructions performed annually in the U.S.
In the 2013 National Football training camps, more than a dozen players were sidelined with ACL injuries. This spate of ACL tears is sure to ripple through high school and college football and soccer fields this fall.
A complete ACL tear is a devastating injury for athletes, typically ending the player’s season and requiring surgical reconstruction. Although many athletes return to the field after reconstruction and physical therapy, studies suggest as many as 80 percent will develop arthritis within 14 years of the injury.
Moreover, children and adolescents are not considered good candidates for ACL reconstruction. The conventional procedure requires surgeons to drill tunnels through the growth plates—the developing cartilage near the end of long bones—but this can disrupt bone growth.
Boston Children’s Hospital orthopedic surgeon Martha M. Murray, MD, wants to change the game plan for ACL injuries. Her research focuses on bio-enhanced ACL repair that uses a bio-engineered scaffold saturated with the patient’s own blood to stimulate healing and to promote clotting, Full story »
Thorsten Schlaeger, PhD, heads the Human Embryonic Stem Cell Core of the Boston Children’s Hospital Stem Cell Program.
I recently took my 6-year-old son to a Family Science Day, hosted by the 2013 American Association for the Advancement of Science (AAAS) Annual Meeting in Boston. He was most excited by a model airplane made out of parts that had been generated with a 3D printer. The scientist, from MIT, explained to us how this technology works: Instead of generating 2D printouts by spraying ink onto paper, 3D printing technologies assemble 3D objects layer by layer from a digital model, generally using molten plastics or metals.
3D printing is quickly being adopted by many professions, from architects and jewelers who want to build mock-ups for clients, to manufacturers of products like bikes, cars or airplanes. Soon we might all have 3D printers in our homes: The kids could design and print their own toys, while the grownups might use the technology to generate replacement parts for minor home improvement jobs (our broken shower faucet knob comes to mind). Full story »

We're at the cusp of integrating miniaturized electronics and monitoring into engineered tissues and organs.
At the start of the 2009 Star Trek reboot (this is relevant, trust me), the USS Kelvin’s captain meets the enemy on their ship to try to negotiate a cease-fire. His crew uses a kind of sensing technology to track his vital signs—like heart rate, breathing, body temperature—right up to the moment of his untimely demise.
While we’re not quite up to the technology level of the Star Trek universe, the ability to remotely sense what’s going on in tissues and organs is something of a holy grail for bioengineers. This is especially true for artificial or engineered organs: If you’d grown a new kidney for a patient needing a transplant, for example, you’d want some way to monitor it and make sure it’s working properly. It’s something that the body does naturally, but that bioengineers have struggled to replicate. Full story »
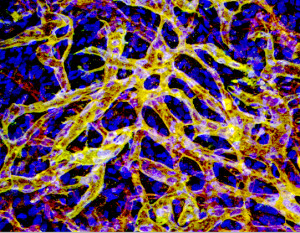
Within days of injecting a cell mix into mice, numerous blood vessels form. Can these vessels be made to secrete drugs, without the need for IVs or injections?
People who rely on protein-based drugs often have to endure IV hookups or frequent injections, sometimes several times a week. And protein drugs – like Factor VIII and Factor IX for patients with hemophilia, alpha interferon for hepatitis C, interferon beta for multiple sclerosis — are very expensive.
What if they could be made by people’s own bodies?
Combining tissue engineering with gene therapy, researchers at Children’s Hospital Boston showed that it’s possible to get blood vessels, made from genetically engineered cells, to secrete drugs on demand directly into the bloodstream. They proved the concept recently in the journal Blood, reversing anemia in mice with engineered vessels secreting erythropoietin (EPO).
This technology could potentially deliver other protein drugs, Full story »
How do cells figure out how to build three-dimensional organs with multiple kinds of tissues? A group of engineers, geneticists, biochemists and cell biologists at Children’s Hospital Boston and Harvard’s Wyss Institute for Biologically Inspired Engineering sunk their teeth into this mystery – starting, in fact, with the tooth.
Scientists have known for more than a century that the growth of many organs (including tooth, cartilage, bone, muscle, tendon, kidney and lung) begins with the formation of a compact cell mass called “condensed mesenchyme.” But what makes this mass form to begin with? Until now, no one knew. Full story »
People who have had a heart attack or have coronary artery disease often sustain damage that weakens their heart. Milder forms of heart failure can be treated with medications, but advanced heart dysfunction requires surgery or heart transplant. A team of physicians, engineers and materials scientists at Children’s Hospital Boston and MIT offers two alternative ways to strengthen weakened, scarred heart tissue — both involving nanotechnology.
One approach blends nanotechnology with tissue engineering to create a heart patch laced with gold whose cells all beat in time – as shown in the above video.
The other uses minute nanoparticles that can find their way to dying heart tissue, carrying stem cells, growth factors, drugs and other therapeutic compounds. Full story »
A patient's own cells may be able to create fat tissue with its own blood supply. (Image: Jagiellonian University Medical College)
The majority of the millions of plastic surgeries performed in the U.S. each year aren’t cosmetic procedures for Hollywood starlets or Beverly Hills housewives trying to hold on to their youthful looks. They’re reconstructive operations for patients with disfiguring injuries, tumor resections and congenital defects such as childhood hemangiomas, which can occur on the face.
A big challenge in reconstruction is compensating for the loss of a large volume of subcutaneous fat. Currently, there are three ways to do this, none of them ideal. Full story »