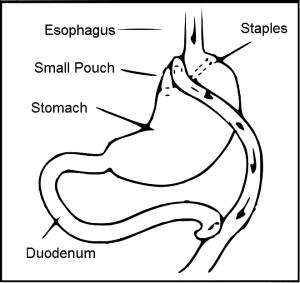
Gastric bypass surgery creates a small pouch in the stomach and connects it directly to the small intestine. Why does it help type 2 diabetes? (Wikimedia Commons)
But why? To investigate, a research team led by Nicholas Stylopoulos, MD, of Boston Children’s Hospital’s Division of Endocrinology, spent a year studying rats and observed that after gastric bypass surgery, the way in which the small intestine processes glucose changes. They saw the intestine using and disposing of glucose, and showed that it thereby regulates blood glucose levels in the rest of the body, helping to resolve type 2 diabetes.
Basically, as the team reported recently in Science, the small intestine—widely believed to be a passive organ—is actually a major contributor to the body’s metabolism.
Gastric bypass surgery, typically reserved for severely obese patients, reroutes food into the smaller pouch of the stomach and bypasses the rest of the stomach and duodenum.
“We have seen type 2 diabetes resolve in humans after gastric bypass, but have never known why,” says Stylopoulos. “People have been focusing on hormones, fat and muscle, but this study has shown that the answer lies somewhere in the small intestine most of the time.”
This PET/CT scan shows the intestine of a rat taking up more glucose after gastric bypass surgery—note the increased yellow and orange in the intestines in the rat on the right, as compared with the “before” image at left.
“Previously, we had not considered the intestine as a major glucose-utilizing organ. We have found this process is exactly what happens after surgery,” says Stylopoulos.
Type 2 diabetes resolved in 100 percent of the rats that underwent gastric bypass in the study. Sixty-four percent of type 2 diabetes cases was resolved by the intestine, and the researchers hypothesize that the other 36 percent may have resolved due to weight loss or other factors.
These findings, featured in the NIH Director’s blog, pave the way for investigations of how to get the intestine to burn sugar without the surgery. “With further research, we may find ways to bypass the bypass,” says Stylopoulos. “The results of our study are promising because, unlike the brain and other organs, intestines are easily accessible. Also, since cells in the intestine have such a short lifespan, we can easily study and pharmacologically manipulate them to use glucose, without long-term problems.”
Citation:
![]() Saeidi N, Meoli L, Nestoridi E, Gupta NK, Kvas S, Kucharczyk J, Bonab AA, Fischman AJ, Yarmush ML, & Stylopoulos N (2013). Reprogramming of intestinal glucose metabolism and glycemic control in rats after gastric bypass. Science (New York, N.Y.), 341 (6144), 406-10 PMID: 23888041
Saeidi N, Meoli L, Nestoridi E, Gupta NK, Kvas S, Kucharczyk J, Bonab AA, Fischman AJ, Yarmush ML, & Stylopoulos N (2013). Reprogramming of intestinal glucose metabolism and glycemic control in rats after gastric bypass. Science (New York, N.Y.), 341 (6144), 406-10 PMID: 23888041







